The UK-based charity said just because a product is BPA-free does not mean there are no health risks and authorities should regulate groups of related chemicals instead of taking a substance-by-substance approach.
It added some firms are claiming, when selling to downstream users, that BPS has no hazards.
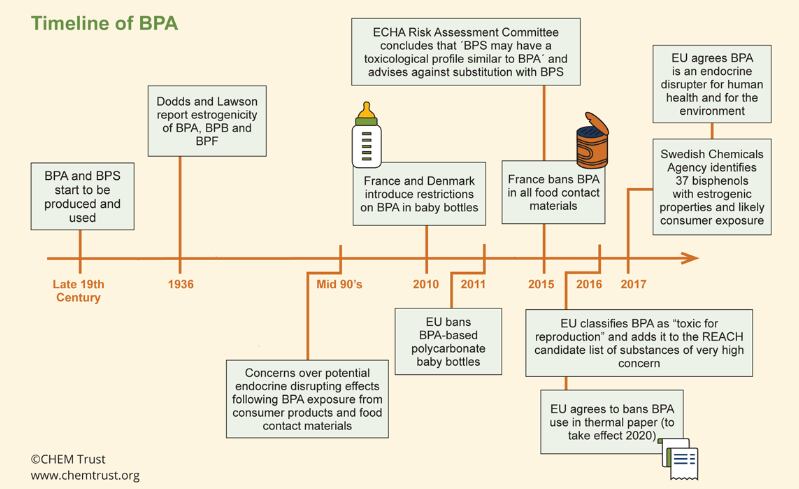
European Commission regulation means stricter measures on bisphenol A (BPA) will apply from September this year.
Migration limit has been strengthened 12-fold from 0.6 mg/kg to 0.05 mg/kg in food contact plastics and it will be prohibited in food contact varnished or coated materials for young children.
The European Food Safety Authority (EFSA) is re-evaluating BPA with results expected in 2019.
Substitution situation ‘not acceptable’
CHEM trust said companies have moved to other bisphenols such as bisphenol S (BPS), bisphenol F (BPF) and bisphenol HPF (BHPF).
“When looking for a new chemical to use in an application, companies will look for one of a similar structure, as it is likely to have similar properties. However, this similarity often extends to toxicity, which is what the scientists are finding with bisphenols,” it added.
“Despite the EU’s sophisticated chemicals regulatory systems, industry has been permitted to replace one worrying bisphenol with another. This is not an acceptable situation, as the health of future generations is at stake.”
Dr Michael Warhurst, executive director of CHEM Trust, said: “This report shows that people and the environment are not being properly protected from hazardous chemicals as businesses are moving from one problem chemical in a group to another.
“We need EU regulators to phase out groups of chemicals of concern, rather than slowly restricting one chemical at a time. We cannot continue to gamble with people’s health like this.”
Call to restrict groups of similar chemicals
CHEM Trust has written to ECHA, EFSA and the European Commission’s Health Commissioner, to ask them to restrict use of groups of similar chemicals to prevent substitution of one hazardous chemical with ones that have similar properties.
It said the only exception should be if industry has data showing the chemical they wish to use does not have the same properties as the one being restricted.
The European Commission previously found most food and beverage cans use BPA based epoxy-resin technology as a coating.
The metal can manufacturing sector has said switching to alternatives would mean a decrease in the shelf life of canned food by one to two years which could lead to an increase in food waste.
Dr Paloma Alonso Magdalena, associate professor of Nutrition at Miguel Hernández University of Elche (UMH), Spain, who reviewed the CHEM Trust report, said: “BPA is now well known to have endocrine disrupting properties, affecting a wide range of the body’s systems, from reproductive organs to our metabolism.
“It is very worrying that a number of other bisphenols are now being found to have similar hazardous properties. It does not seem wise to be moving from BPA to these chemicals.”
CLARITY-BPA and BPA detection by Vicam
The US Food and Drug Administration (FDA) recently reasserted its position the chemical is not harmful. This was based on a National Toxicology Program (NTP) pre-peer review draft report.
The Endocrine Society said the conclusion was ‘premature’ and judgement should be reserved until the full report in 2019.
The Can Manufacturers Institute (CMI) claims more than nine out of 10 food cans sold in the US are lined with coatings not derived from BPA and are made from acrylic and polyester material.
Meanwhile, Vicam has introduced a method for determination of BPA for food and agricultural labs.
The Waters Corporation business said BPATest isolates and purifies BPA for analysis from 0.1 to 50 parts per billion in ten minutes of preparation time.
BPATest pairs immunoaffinity chromatography with liquid chromatography (HPLC, UPLC) and fluorescence or mass spectrometry detectors.
The proprietary monoclonal antibody in Vicam’s immunoaffinity columns transforms test samples into highly concentrated BPA extract.
Vicam claimed the quantitative method outperforms solid phase extraction and ‘dilute-and-shoot’ techniques in sensitivity and specificity.
Marjorie Radlo-Zandi, MD at Vicam, said: “Worldwide efforts to minimize the impact of BPA on the population, consumer confidence and trade revenues continue to evolve toward even tighter control measures worldwide. BPATest serves laboratories in their pursuit of analytical excellence in empowering food manufacturers and regulators to safeguard human health.”
